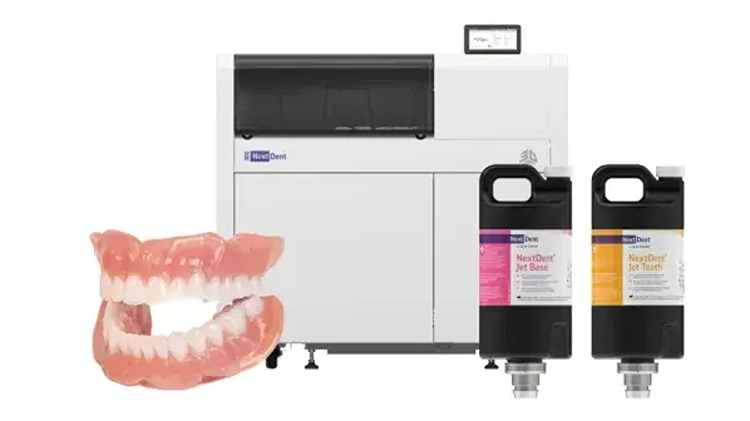
ROCK HILL, SC, Apr 29, 2026 – 3D Systems has received EU Class IIa certification for its NextDent Jet Base and NextDent Jet Teeth materials, clearing the NextDent 300 MultiJet printer workflow for commercial denture production across the European Union.
The certification covers the complete NextDent Jetted Denture solution under Medical Device Regulation (MDR) 2017/745. It follows 3D Systems’ full-scope EU MDR certification, which the company reported in March 2026.
The MDR framework sets quality, safety, and clinical evidence requirements for moderate-risk medical devices sold in the EU. The NextDent Jetted Denture solution uses the NextDent 300 printer with the Jet Base and Jet Teeth materials to produce patient-specific monolithic dentures in a single print.
The dentures are cured after printing and ready for finishing without additional post-curing steps. 3D Systems described the workflow as a monolithic, multi-material jetted denture process for denture applications within its dental portfolio, which includes straighten, protect, repair, and replace.
Jeffrey Graves, Ph.D., president and chief executive officer of 3D Systems, stated: “Achieving Class IIa MDR certification for our NextDent Jetted Denture Solution is a major milestone that validates the strength of our clinical data, quality systems, and innovation pipeline. By launching in Europe on May 4, well ahead of our original summer timeline, we are immediately expanding access to this transformative technology for dental labs and clinics across the region. This clearance substantially increases our available addressable market for this high-value platform. As adoption accelerates in both the U.S. and Europe, we expect it to drive significant recurring revenue through premium materials while reinforcing our position as the leading full-service provider in digital dentistry.”
3D Systems cited market data that estimate the European denture segment as a multi-billion-dollar opportunity. With the certification, the company can supply EU customers with a regulated denture production workflow that includes the NextDent 300 MultiJet printer, certified NextDent Jet Base and NextDent Jet Teeth materials, software, and application support. Commercial availability in EU markets begins May 4, 2026, through 3D Systems and its authorized distribution partners.
Source: 3D Systems
About 3D Systems
![]()
3D Systems was founded in 1986 by Charles “Chuck” Hull, the inventor of stereolithography (SLA) and the first commercial 3D printer, the SLA-1. Based in Rock Hill, SC, the company has played a central role in the development of additive manufacturing technologies. Today, 3D Systems provides a range of 3D printing solutions that include hardware, software, and materials. Its offerings support a wide variety of industries – such as aerospace, automotive, healthcare, dental, and consumer goods – enabling workflows that range from early-stage prototyping to full-scale production. Its infrastructure spans offices, manufacturing facilities, and Customer Innovation Centers designed to support integration of 3D printing into industrial and commercial operations. Over the decades, 3D Systems has evolved from a technology originator into a solutions provider, focusing on application-specific capabilities across sectors including medical and dental, aerospace and defense, transportation, AI infrastructure, and industrial goods. Its engineering teams focus on improving manufacturing outcomes using additive technologies.
