A new study shows that densifying a ceramic solid electrolyte called argyrodite (Li₆PS₅Cl) can dramatically reduce the risk of lithium dendrite formation in all-solid-state batteries. As battery makers seek safer, more energy-dense alternatives to liquid electrolytes, solid versions hold promise, but dendrites (needle-like lithium deposits) remain a big barrier, because they can pierce the solid medium and cause short circuits, tells Tech Xplore.
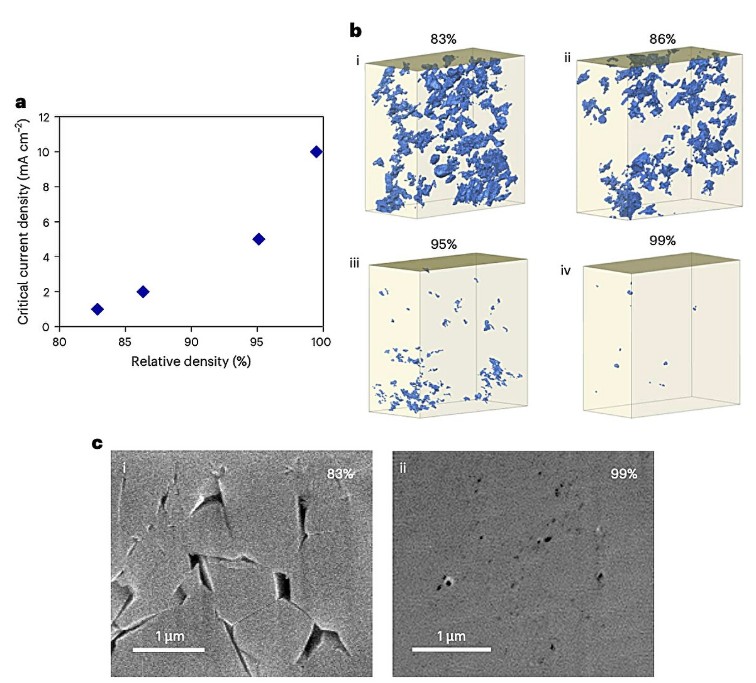
In their experiments, the researchers increased the relative density of Li₆PS₅Cl from about 83% (cold-pressed) up to 99% using spark plasma sintering at varied temperatures. They then studied how the microstructure (pores, cracks, voids) shifted with densification and how these changes affected how much current the electrolyte could support without dendrite growth.
The key result: at 99% density, the material tolerated lithium plating currents as high as 9 mA per square centimeter without forming dendrites. That’s a major leap over prior limits. The authors link the improvement to reductions in pore size and crack length, microstructural features that, when minimized, help maintain a uniform current distribution and prevent lithium from finding weak paths.
Modeling also shows that not every structural change helps. While smaller pores and shorter cracks raise the “critical current density” threshold, reducing the number of pores or narrowing cracks too much can have counterintuitive drawbacks. Yet in their tests, the beneficial effects dominated.
This advance doesn’t fully solve all challenges for solid electrolytes, i.e., issues such as long-term stability, interfaces to electrodes, and scale remain. But it offers a clear path: pushing toward near-full density in argyrodite and similar materials could make solid batteries safer and more practical. Future work will need to integrate these electrolytes into full cells and verify performance under real cycling conditions.
Densifying ceramic electrolytes could be a turning point. It shows that the right micro-architecture may tame dendrites and bring solid batteries closer to real applications.
